Travel nursing jobs are seeing some cooling down after several years of high demand and high rates, but here are some of the highest-paid travel nursing jobs available across the country.
New travel nursing jobs available. Speak with a recruiter today.
Travel nursing during COVID
As the Nejm Catalyst Innovations in Care Delivery journal reported in October 2020, the first wave of COVID-19 resulted in an increased demand for ICU travel nurses in hot spots like Massachusetts and New York — available contracts were up 612% and 1,038% respectively. During the second wave, available ICU travel nurse jobs continued to climb, including an increase of 732% in Arizona and by 927% in Texas.
As you can imagine, pay for travel nurses also increased. The journal found that by April, the average weekly pay for ICU travel nurses in New York had increased by as much as 74%. Massachusetts saw an average weekly salary increase of 39%, while Florida nurses’ weekly salary went up by 33%. By July, Arizona saw an increase of 44% while Florida demand increased by 61%.
Overall, if you were a travel nurse who had any interest in working in a COVID unit or ICU during the past year, you had ample opportunity to find a job and make significantly more money than in the past.
For some travel nurses, it was a chance to expand into a new field and try something new. For others, it was an opportunity to take a job that held deep meaning and help in a time of crisis. But not everyone needs to be a COVID nurse.
COVID has actually created job opportunities outside of the acute care setting and, in states not experiencing outbreaks, regular travel nurse assignments are still available.
Non-COVID nurse travel jobs
The challenge with finding non-COVID travel nurse jobs is many hospitals in hot spots have cancelled or reduced elective procedures and non-essential medical services. As a result, many staff nurses have been floated or laid off and there’s simply less demand for travel nurses at those facilities.
But COVID itself has created some additional opportunities for travel nurses, including:
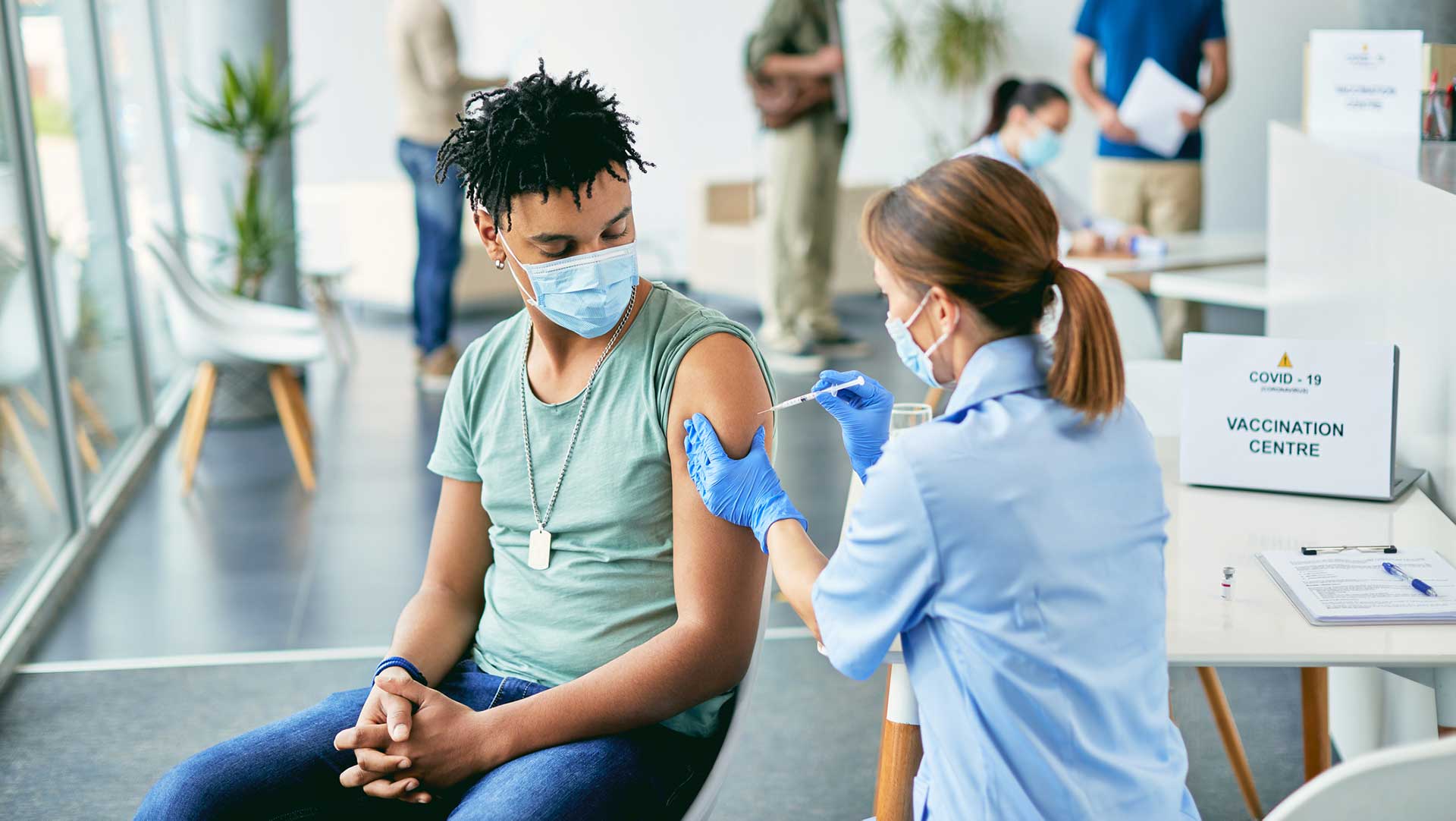
Vaccine clinic nurses
From mass vaccination clinics to health departments to private facilities offering vaccinations, many are turning to travel nurses to fill these temporary roles. RNs are vital to vaccination clinics where they do everything from educating, screening patients, administering the vaccine and monitoring patients for any reactions after injection.
Travel nurses are ideal vaccine clinic RNs because these are temporary positions and staffing needs are variable. They’re also a good opportunity for LPNs to take on temporary assignments too. For travel nurses who are looking for positions that keep them out of the ICU, a clinic can be an ideal environment. Plus, as someone with experience working as a vaccine nurse myself in a volunteer capacity, I can confirm it’s also an incredibly rewarding and fun place to be.
The pressing need for vaccine nurses has slowed as vaccination rates overall in the country have decreased. Currently, there aren’t a lot of specific positions for travel vaccine nurses, although that could change as numbers increase and flu season begins.
Ready to start travel nursing? Talk to a recruiter today.
ER nurses
Here is a sampling of ER travel nurse jobs available:
- California: $4.7K/week
- Massachusetts: $5.7K/week
- Minnesota: $3.7K/week
- New Jersey: $4.6K/week
- New York: $5.4K/week
- North Carolina: $4.4K/week
- North Dakota: $2.8K/week
- Wisconsin: $3.3K/week
OR nurses
Here is a sampling of OR travel nurse jobs available:
- California: $3.6K/week
- Colorado: $3K/week
- Massachusetts: $4.8K/week
- Michigan: $4.2K/week
- Minnesota: $2.9K/week
- Missouri: $3K/week
- Montana: $3K/week
- New Jersey: $3.4K/week
- New York: $2.6K/week
- Tennessee: $2.7K/week
- Texas: $4.8K/week
- Virginia: $3.8K/week
- Washington: $5.4K/week
- West Virginia: $4K/week
Hemodialysis nurses
There has been a steady stream of hemodialysis travel nurse positions throughout the pandemic and many of them come with high pay.
For instance, here are the rates currently available for hemodialysis positions right now:
- Massachusetts: $1.2K/week
- Michigan: $2K/week
- South Dakota: $2.3K/week
Labor and delivery nurses
L&D travel nurse jobs available include the following:
- California: $5.9K/week
- Kansas: $2.9K/week
- Missouri: $4.5K/week
- New York: $5.3K/week
- Oregon: $4.1K/week
- Pennsylvania: $4.2K/week
- Washington: $4.7K/week
Infusion nurses
Infusion nursing is also a specialty that has seen increased demand since the beginning of the pandemic. Although infusions can be done for a variety of medical conditions, antibody infusion therapy is increasingly being offered for COVID-19 patients. It’s sometimes offered as an outpatient treatment, meaning you won’t necessarily need to be on an actual COVID unit in order to be an infusion nurse.
Here are the salaries currently available for open infusion nurse positions:
- Iowa: $2.7K/week
- Michigan: $2.8K/week
- Missouri: $2.8K/week
- Oregon: $2.8K/week
- Vermont: $3.1K/week
Behavioral health nurses
The following pay rates are currently available for psychiatric RN-specific positions:
- New York: $3.4K/week
- Pennsylvania: $3.6K/week
Office nurse
How about a nice relaxing stint as an office nurse or an outpatient clinic RN? Search for “outpatient travel nurse jobs” or “office nurse travel jobs” to find some in your area of choice.
Med/Surg nurses
You’ll find a wide variety of openings for Med/Surg nurses across the country. We found the following jobs open right now:
- Alabama: $2.5K/week
- California: $4.7K/week
- Georgia: $3.2K/week
- Indiana: $3.8K/week
- Massachusetts: $5.1K/week
- Michigan: $4.3K/week
- Missouri: $3.7K/week
- Nebraska: $3.2K/week
- New Hampshire: $4.3K/week
- New Jersey: $4K/week
- New York: $4.8K/week
- Pennsylvania: $3.2K/week
- Wisconsin: $3K/week
- Wyoming: $3.2K/week
Pediatric
Here are some of the highest-paying PICU and PCICU travel nursing assignments we found across the country:
- California: $6.1K/week
- District of Columbia: $6.3K/week
- Florida: $3.3K/week
- Massachusetts: $5.2K/week
- Minnesota: $6.1K/week
- Missouri: $4.6K/week
- Nevada: $5K/week
- New Jersey: $5.4K/week
- New York: $4K/week
- North Dakota: $4.3K/week
- Texas: $4.7K/week
ICU
In general, ICU travel nurses have the opportunity to make the most money in the following locations:
- California: $4.8K/week
- Idaho: $4.6K/week
- Massachusetts: $4.6K/week
- Michigan: $3K/week
- New Hampshire: $4.4K/week
- New Jersey: $4.4K/week
- New York: $4.9K/week
- Wisconsin: $4.5K/week
Telemetry Nurses
If your specialty is in telemetry, consider these travel assignments:
- California: $4.8K/week
- Indiana: $3.8K/week
- Massachusetts: $3.1K/week
- New Jersey: $4.5K/week
- Oregon: $4.2K/week
- Rhode Island: $4.6K/week
Other Specialties
For niche nursing assignments, here are some of the highest-paying one-off opportunities:
- Endoscopy/GI (New York): $5.6K/week
- Interventional Radiology (New York): $6K/week
- MedSurg/TM (Pennsylvania): $5.7K/week
- Pediatrics (New York): $5.1K/week
Non-COVID travel nursing positions
If you’re a travel nurse looking to avoid the COVID unit — or even if you’re a COVID nurse who needs a break to recuperate from burnout — there are plenty of opportunities available to be a travel nurse during the pandemic.
New travel nursing jobs available. Speak with a recruiter today.





